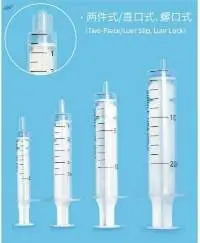
Size: 1 ml, 3ml, 5ml, 10ml, 20ml, 50ml - Nieedle Size: 26G x y2 25G x I ,)-3 G x I , 22G X I 1 l4 , ZlCi x 1 112", 20G x I 1pc/polybag or 1pc/blister bag disposable syringe 1ml:3000pcs/48.5*40.5*43.5cm 2/3ml:3200pcs/61.5*48.5*38cm 5ml:2400pcs/61.5*48.5*38cm 10ml:2400pcs/61.5*48.5*38cm 20ml:1800pcs/61.5*48.5*38cm 30ml:1200pcs/61.5*48.5*38cm 50/60ml:800pcs/53*47.5.45.5cm
Disposable Syringes 3ml 3Part Hypodermic Syringe Sterile , with Needle Size : 23 G x 1 It is suitable for subcutaneous, intradermal, intramuscular, intravenous injection or drainage. Material-medical grade PP Color-transparent Certificate -CE, ISO Specification -1ml, 2ml, 3ml, 5ml, 10ml, 20ml, 30ml, 50ml
-Key Specifications/Special Features -15 different types of 1ml, 2ml, 3ml, 5ml,10ml... Price: 0.03$/pcs *1ml with needle, Luer Slip Payment: TT20%-LC80% * The price depends on the types * Price is FOB Vietnam Process / Procedure: 1. Buyer send LOI 2. Seller provide certificates and FCO 3. Buyer sign FCO 4. Sign SPA 5. Buyer pay deposit 6. SGS Inspection 7. Loading / delivery 8. Buyer pay balance
Anti-disable Syringe with needle for Fixed-Dose Immunization With Self-destructive characteristics, the self destructive performance of the syringe will begin to take effects as the injestion starts. When the fixed dose of vaccine is finished, the syringe will be completely sell-locked, effectively preventing the reuse of the syringe and improving the safety of madical injection.
Disposable Insulin Syringe with Needle The integrated design of the needle holder and barrel injection molding which ensures the low residue of the liquid and ensures the accuracy of the injection of the liquid. Excellent sliding performance, convenient for clinical injection operation medical staff.
Auto Diable self-destructive  Syringe with needle It has self-destructive charcateristics. When the expected dose of the drug is injected, the self-destructive performance will automatically take effect, effectively prevent the reuse of syringes, avoild cross-infection and improve the safety of clinical injection.
Profhilo 64mg Syringe (1x2ml) Princess Rich (1x1.0ml) Princess Filler (1x1.0ml) Princess Volume (1x1.0ml) Revanesse Pure (2x1ml) Revanesse (2x1ml) Revanesse Ultra (2x1ml) Revanesse Lips (2x1ml) Redexis (2x1ml) Redexis Ultra (2x1ml) Stylage Hydro (1x1.0ml) Stylage Hydro Max (1x1.0ml) Belotero Basic (1x1.0ml) Belotero Intense (1x1.0ml) Belotero Soft (1x1.0ml) Radiesse 1.5ml Radiesse 0.8ml Serious Inquiries ONLY Contact for more information
Excellent slidable performance for medical staff to easily perform clinical injection. Strong graduation ink adhesive force, friction resistance, not easy to fall down. Universal fit: Conical lock fittings with 6% confirm with international standards, can be used with any product with 6% conical lock fittings. Clearly observe solution and bubbles with transparent barrel, push button of plunger with skidproof structure that easy to grip, the inner cavity of the rear end of barrel with skidproof structure to prevent the plunger from accidentally sliding. Three-piece, Two-piece Luer Slip, Luer Lock Concentric, Excentric. It can meet different needles.
Sterile syringes in USA from manufacturer's warehouse. Shipment every week. Country of origin: China. FDA/CE approved. Prices OTG LA: 1ml luer slip with 25gx1''needle 3ml luer lock with 23dx1'' needle 1ml luer slip US$0.60 3ml luer lock US$0.65 Also I'm sending you edited documents for your customer's reference. FOB price US$0.30 60 million OTG in LA every week
Insulin Syringe EO Sterilization Refer Standards ISO 9626: 1991 + AMENDMENT 1 2001 Stainless steel needle tubing for the manufacture of medical devices. ISO 8537:2007 Sterile single-use syringes, with or without needle, for insulin. ISO 10993-7:2008 Biological evaluation of medical devices - Part 7 : Ethylene oxide sterilization residuals. ASTM F 1140/F 1140M-13 Standard test methods for internal pressurization failure resistance of unrestrained packages. USP 37-NF 32 Bacterial Endotoxins Limit. ISO 11737-2:2009 Sterilization of medical devices - Microbiological methods - Part 2: Test of sterility performed in the definition, validation and maintenance of a sterilization process. ISO 11135-1:2007 Sterilization of health care products - Ethylene oxide - Part 1: Requirements for development, validation and routine control of a sterilization process for medical devices.
Safety Slide Insulin Syringe EO Sterilization Refer Standards EN ISO 8537:2007 Sterile single-use syringes, with or without needle, for insulin. EN ISO 9626:1991 / AMD. 1: 2001 Stainless steel needle tubing for the manufacture of medical devices AMENDMENT 1. ASTM F88/F88M-09 Standard Test Method for Seal Strength of Flexible Barrier Materials. ASTM F1140/F1140M -13 Standard Test Methods for Internal Pressurization Failure Resistance of Unrestrained Packages. ISO 10993-7:2008 Biological evaluation of medical devices - Part 7: Ethylene oxide sterilization residuals. USP 37- NF32 2014: U.S. Pharmacopeia National Formulary.