Item condition: second hand Interchange Part Number: 2011 2012 2013 2014 2015 2016 2017 2018 11 12 13, CHRYSLER AWD ALL WHEEL DRIVE SRT-8 8 MOPAR, P53022071GB, 53022071 Manufacturer Part Number: R8193010AA Number of Cylinders: 8 Engine Size: 6.4L Fuel Type: Gasoline Brand: FACTORY ORIGINAL GENUINE JEEP SRT8 8 4 DOOR SUV Fitment Type: Direct Replacement Mileage: 71K Warranty: 60 Day UPC: Does not apply Items Size Qty Box  Dimensions 22X11X7.3 cm 100 Pcs/Box Carton Dimensions 38 X23.5 X 23.5 cm 1000 Pcs/Carton=10 Boxes Total Weight/Carton S:4.6kg M:4.8kg L:5.3kg XL:6.0kg Container Cartons Boxes Pcs 1x 20GP 1400 Cartons 14,000 Boxes 1,400,000 Pcs 1 x 40GP 2600 Cartons 26,000 X ½ X Boxes 2,600,000 Pcs 1x40 HQ 3233 Cartons 32,330 Boxes 3,233,000 Pcs Certificates & Test Reports All our Certifications & Tests are Real No. Items Certifications & Tests 1 Factory Quality Certifications ISO13485, ISO9001, BSCI 2 EU Markets EN455 1-3 Test, DOC - EU Registration, German DIMI, EN374 1-5 Test EN374 CEEN21420 - EN16523 3 US Markets U.S. 510K, FOOD Grade Test, Reach Test RoHS,ASTM D6978, ASTM D6319 4 Others Acute Systemic Toxicity Test, Skin Irritation Test, Skin Sensitization Test, GB10213, GB4806

Item details Item status : second hand Seller notes : CONDITION: REMANUFACTURED BLOCKENGINE WAS RUNNING GREAT BLOCK IS BARLEY USED REMANUFACTURED IN 6/22/2020, TO WATCH THE CAR RUNNING VIDEO LINK IS BELOW...." Item condition: second hand Interchange Part Number: 2011 2012 2013 2014 2015 2016 2017 2018 11 12 13, CHRYSLER AWD ALL WHEEL DRIVE SRT-8 8 MOPAR, P53022071GB, 53022071 Manufacturer Part Number : R8193010AA Number of Cylinders : 8 Engine Size: 6.4L Fuel Type : Gasoline Brand : FACTORY ORIGINAL GENUINE JEEP SRT8 8 4 DOOR SUV Fitment Type: Direct Replacement Mileage : 71K Warranty: 60 Day UPC : Does not apply Items Size Qty Box Dimensions 22X11X7.3 cm 100 Pcs/Box Carton Dimensions 38�?23.5�?23.5 cm 1000 Pcs/Carton=10 Boxes Total Weight/Carton S:4.6kg M:4.8kg L:5.3kg XL:6.0kg Container Cartons Boxes Pcs 1x 20GP 1400 Cartons 14,000 Boxes 1,400,000 Pcs 1 x 40GP 2600 Cartons 26,000 Boxes 2,600,000 Pcs 1x40 HQ 3233 Cartons 32,330 Boxes 3,233,000 Pcs Certificates & Test Reports All our Certifications & Tests are Real No. Items Certifications & Tests 1 Factory Quality Certifications ISO13485, ISO9001, BSCI 2 EU Markets EN455 1-3 Test, DOC EU Registration, German DIMI, EN374 1-5 Test EN374 CE EN21420 EN16523 3 US Markets U.S. 510K, FOOD Grade Test, Reach Test RoHS,ASTM D6978, ASTM D6319 4 Others Acute Systemic Toxicity Test, Skin Irritation Test, Skin Sensitization Test, GB10213, GB4806
- Protection Level: EN13795, AAMI Level 2, AAMI Level 3 - Fabric Options: Alcohol Repellent Anti-Static SMMMS Nonwoven (or) Anti-Static SMMMS Nonwoven - Packed with 2 Hand Towels, Lint Free, Scrim Reinforced - Pouched in MGP Pouch - EO Sterile - Third Party Report for AAMI Protection & EN13795 Standard: Available
- Compliant to ASTM F1670 (Blood Penetration Resistance) & ASTM F1671 (Viral Penetration Resistance) standards - at Fabric & at Critical Areas - Fabric Used: Breathable Trilaminate Nonwoven - Packed with 2 Hand Towels - Lint Free - Scrim Reinforced - Pouched in MGP Pouch - EO Sterile - Reports & Batch Records shall be provided - 13485 MDSAP Audited Facility - Third Party Report for AAMI Protection Level 4: Available (Intertek)
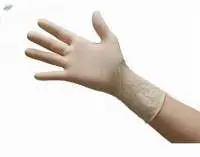
High quality medical grade gloves made from low allergic natural rubber latex, rigorously inspected and tested to exceed the requirements of the European Specification EN455 Part 1 (Freedom from holes), EN455 Part 2 (Physical Properties) and EN455 Part 3 (Biocompatibility) and ASTM D-3578. Lightly powdered with corn starch for bio-absorbability. * Natural Rubber Latex * Ambidextrous * Manufactured in Accordance with ASTM D3578/ EN 455/ FDA Specifications * Maximum Comfort and Protection * Lightly Powdered with USP Absorbable Dusting Powder * Fitting Beaded Cuff for easy donning and to prevent roll down * Available in a smooth surface or an exclusive textured surface * High tensile strength to minimize tearing
SteriMed Powderfree Latex Examination Gloves provide yet another complimentary alternative to meet the stringent demands of diverse users Ideal for medical use these gloves undergo an advanced halogenation washing process which minimizes sensitivity to allergic reactions * These lowprotein gloves also feature a specially textured surface to improve grip Pigment free with no fillers or waxes they offer better protection as they are less likely to absorb body fluids * Natural Rubber Latex * Ambidextrous * Manufactured in accordance with ASTM D3578 EN 455 FDA specifications * Low levels of soluble protein to further reduce incidences of skin sensitivity reactions * Micro roughened nonslip surface for optimal sensitivity * Fitting Beaded Cuff for easy donning * Continuous polymer coating buffers wearer from direct contact with latex to further reduce the risk of skin irritation
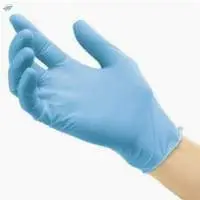
SteriMed Nitrile Gloves contain butadieneacrylonitrile a synthetic copolymer containing no natural rubber latex protein This eliminates the potential for Type I allergic reactions among NRL sensitive individuals and is chemically toughened for resistance in most solvents including petroleum * High flexibility and comfort * Excellent resistance against punctures water permeability * Superior chemical and solvent barrier protection * Textured fingertips for wet and dry grip * Fitting Beaded Cuff for easy donning and to prevent roll down * Excellent tactility with minimized hand fatigue
SteriMed Sterile Surgical Gloves are anatomically designed to provide maximum comfort and grip while reducing hand fatigue to the user The extractable NR latex proteins content is kept to the minimum thereby reduce skin allergic reactions In addition the gloves contain moderate dusting powder level which facilitates swift removal of powder prior to surgical procedures * High Strength Natural Rubber Latex * Low Modulus * Maximum Comfort and Protection * Micro roughened nonslip surface for excellent grip * Softer latex reduces hand fatigue and stronger latex reduces glove tearing * Conform to ASTM D3577 and EN 455 Specifications * Hypoallergenic minimizing potential allergic risks * Sterilized by Gamma Irradiation * Fitting beaded cuff * Punctureresistant design
* Another alternative to meeting the stringent demands of industry Designed to perform like a premium synthetic glove SteriMed Poly Chloroprene Gloves are without the associated risk of adverse reactions to proteins inherent in natural rubber latex * An effective hand protection barrier against contamination and infectious diseases the glove is also chemically toughened to provide extra protection against a broad range of chemicals and oils making it ideally suitable for handling acids caustics and alcohols * Chloroprene formulation allows for enhanced barrier protection of a synthetic glove with the comfort fit and feel of natural rubber * Being of latexfree nature thus eliminating potential Type I allergic reaction elicited by natural rubber proteins * The formulation offers low modulus allowing greater dexterity tactile sensitivity and reduced hand fatigue * The polymer coating not only facilitates ease of donning but also offers greater resistance to fluid permeation * Ambidextrous with textured fingertips for enhanced wet and dry grip * Low modulus for minimized hand fatigue
1.High-impact protection certified to ANSI Z87.1,Military Requirements and CE EN166. 2.Widest field-of-view and optically correct goggle lens provide unprecedented visual clarity. 3. 2.8 mm polycabonate high impact lens ,High performance U.S. imported coating provides superior protection against fogging and scratching. 4.Top and lateral forced air vents provide constant airflow while filtration media prevents small particle entry. 5.Interchangeable lenses for various light conditions - Clear, Smoke and Yellow 6.Polycarbonate lenses offer 100% protection from UV-A, UV-B and UV-C rays. 7.Pliable frame material and strapping system seals to face comfortably. 8.Low profile design ensures compatibility with helmet,, binoculars and night-vision systems. 9.Optional Prescription (Rx) Carrier available11.WG:8.1KG 12.BOX SIZE38*18*8.5CM 13.CTN SIZE45*40*38CMM 14.PACKING:02 PCS of single goggles in one box,then105 boxes in one CTN1300 PCS/CTN.
1.High-impact protection certified to ANSI Z87.1,Military Requirements and CE EN166. 2. The widest field-of-view and optically correct goggle lens provide unprecedented visual clarity. 3. 2.8 mm polycarbonate high impact lens , High-performance U.S. imported coating provides superior protection against fogging and scratching. 4.Top and lateral forced air vents provide constant airflow while filtration media prevents small particle entry. 5.Interchangeable lenses for various light conditions - Clear, Smoke and Yellow 6.Polycarbonate lenses offer 100% protection from UV-A, UV-B and UV-C rays. 7.Pliable frame material and strapping system seals to face comfortably. 8.Low profile design ensures compatibility with helmet,sights, binoculars and night-vision systems. 9.Optional Prescription (Rx) Carrier available 11.WG:8.1KG 12.BOX SIZE38*18*8.5CM 13.CTN SIZE45*40*38CMM 14.PACKING:02 PCS of single goggles in one box,then105 boxes in one CTN1300 PCS/CTN.
Features: Disposable breathable, soft texture and comfortable to wear Antistatic fashionable and elegant design, durable no stimulus to skin, prevents and isolates dust particle and virus invading. Provides reliable barriers to water stem or blood and other fluids Important to minimize cross-infection during surgery Specifications: Material: nonwoven Colors: blue, white, green, yellow, pink and navy Pattern: long sleeves, knitted cuffs, ties on neck and waist with optional sterilized V-neck and pockets are available. Customized sizes and weights are accepted Size: 115 x 145, 120 x 140 and 130 x 150cm Weight: 22 to 55g Packing: customized