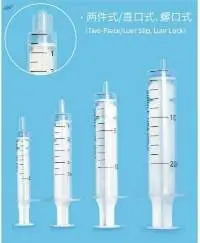
Disposable Syringe 1 C
This disposable syringe 1 C is designed specifically for human subcutaneous and intramuscular injection. In addition, it is made of high molecular weight polypropylene material,and the syringe model can be divided into a syringe and a drug dissolver.
Product Details
Description
This disposable syringe 1 C is designed specifically for human subcutaneous and intramuscular injection. On the other hand, this disposable syringe plays an important role in drug dissolution. Equipped with thick drug dissolver, it can be used for human injection when the fine needle is replaced. In order to better use the post-destruction treatment, the existing self-destructing syringe can not be used again after the use of the needle by retracting the needle, clamping the core rod or automatically breaking the core rod. What's more, the core rod is designed with a self-destructing structure for easy handling after one-time use.
Made of high performance polypropylene, it has good biocompatibility. There is a non-slip structure of the syringe, which brings more convenience for user. It achieves safe injection and easy operation, thus avoiding secondary use in order to eliminate acupuncture infection.
Advantages
1.Really safe injection, eliminating the threat of acupuncture infection
2.Consistent with traditional syringe operating ways, it achieves safe ,easy and convenient injection
Specifications
Model
Disposable syringe1c
Applied Area
Human subcutaneous and intramuscular
Capacity
1c
Applicable sope
Clinic,hospital,healthcare center,and other medical institutions.
Size: 1 ml, 3ml, 5ml, 10ml, 20ml, 50ml - Nieedle Size: 26G x y2 25G x I ,)-3 G x I , 22G X I 1 l4 , ZlCi x 1 112", 20G x I 1pc/polybag or 1pc/blister bag disposable syringe 1ml:3000pcs/48.5*40.5*43.5cm 2/3ml:3200pcs/61.5*48.5*38cm 5ml:2400pcs/61.5*48.5*38cm 10ml:2400pcs/61.5*48.5*38cm 20ml:1800pcs/61.5*48.5*38cm 30ml:1200pcs/61.5*48.5*38cm 50/60ml:800pcs/53*47.5.45.5cm
Excellent slidable performance for medical staff to easily perform clinical injection. Strong graduation ink adhesive force, friction resistance, not easy to fall down. Universal fit: Conical lock fittings with 6% confirm with international standards, can be used with any product with 6% conical lock fittings. Clearly observe solution and bubbles with transparent barrel, push button of plunger with skidproof structure that easy to grip, the inner cavity of the rear end of barrel with skidproof structure to prevent the plunger from accidentally sliding. Three-piece, Two-piece Luer Slip, Luer Lock Concentric, Excentric. It can meet different needles.
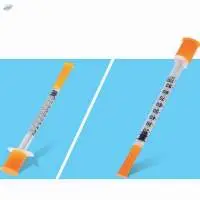
Disposable Insulin Syringe with Needle The integrated design of the needle holder and barrel injection molding which ensures the low residue of the liquid and ensures the accuracy of the injection of the liquid. Excellent sliding performance, convenient for clinical injection operation medical staff.
Disposable Three-Piece,Two-Piece Luer Slip Syringe Luer lock Concentric Excentric Syringe
Disposable Syringes 3ml 3Part Hypodermic Syringe Sterile , with Needle Size : 23 G x 1 It is suitable for subcutaneous, intradermal, intramuscular, intravenous injection or drainage. Material-medical grade PP Color-transparent Certificate -CE, ISO Specification -1ml, 2ml, 3ml, 5ml, 10ml, 20ml, 30ml, 50ml
Supplier: Nitrile/latex/vinyl gloves, masks, surgical gowns, syringes, goggles, ventilator, diapers
Services: Wholesales, medical devices, also provides logistics companies
Buyer: Copper
Insulin Syringe EO Sterilization Refer Standards ISO 9626: 1991 + AMENDMENT 1 2001 Stainless steel needle tubing for the manufacture of medical devices. ISO 8537:2007 Sterile single-use syringes, with or without needle, for insulin. ISO 10993-7:2008 Biological evaluation of medical devices - Part 7 : Ethylene oxide sterilization residuals. ASTM F 1140/F 1140M-13 Standard test methods for internal pressurization failure resistance of unrestrained packages. USP 37-NF 32 Bacterial Endotoxins Limit. ISO 11737-2:2009 Sterilization of medical devices - Microbiological methods - Part 2: Test of sterility performed in the definition, validation and maintenance of a sterilization process. ISO 11135-1:2007 Sterilization of health care products - Ethylene oxide - Part 1: Requirements for development, validation and routine control of a sterilization process for medical devices.