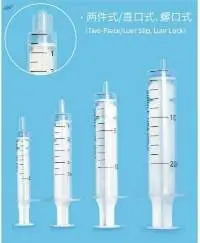
We are a manufacturer for cardiology disposables based in Shenzhen China, we sell control syringe, 10ml or 12ml, The price is competitive and quality is very good.
We are a manufacturer for cardiology disposables based in Shenzhen China, we sell control syringe, 10ml or 12ml, The price is competitive and quality is very good.
Angiography chronary control syringes is made from polycarbonat, high quality, rotatible male luer, 550 psi, finger grip. 50 pcs in bulk ctn.2, 75 kg brutto.
--Smooth operation; --Clear, glass-like poly-carbonate for complete clarity; --Latex free. --Multiple plunger styles. --Non-sterile --Sizes: 10ml, 12ml z
--Smooth operation --Clear, glass-like polycarbonate for complete clarity --Stopper has 0.5ml reservoir --Multiple plunger styles --Non-sterile Size: 10ml/12mlz
Disposable Syringes 3ml 3Part Hypodermic Syringe Sterile , with Needle Size : 23 G x 1 It is suitable for subcutaneous, intradermal, intramuscular, intravenous injection or drainage. Material-medical grade PP Color-transparent Certificate -CE, ISO Specification -1ml, 2ml, 3ml, 5ml, 10ml, 20ml, 30ml, 50ml
Excellent slidable performance for medical staff to easily perform clinical injection. Strong graduation ink adhesive force, friction resistance, not easy to fall down. Universal fit: Conical lock fittings with 6% confirm with international standards, can be used with any product with 6% conical lock fittings. Clearly observe solution and bubbles with transparent barrel, push button of plunger with skidproof structure that easy to grip, the inner cavity of the rear end of barrel with skidproof structure to prevent the plunger from accidentally sliding. Three-piece, Two-piece Luer Slip, Luer Lock Concentric, Excentric. It can meet different needles.
-Key Specifications/Special Features -15 different types of 1ml, 2ml, 3ml, 5ml,10ml... Price: 0.03$/pcs *1ml with needle, Luer Slip Payment: TT20%-LC80% * The price depends on the types * Price is FOB Vietnam Process / Procedure: 1. Buyer send LOI 2. Seller provide certificates and FCO 3. Buyer sign FCO 4. Sign SPA 5. Buyer pay deposit 6. SGS Inspection 7. Loading / delivery 8. Buyer pay balance
Size: 1 ml, 3ml, 5ml, 10ml, 20ml, 50ml - Nieedle Size: 26G x y2 25G x I ,)-3 G x I , 22G X I 1 l4 , ZlCi x 1 112", 20G x I 1pc/polybag or 1pc/blister bag disposable syringe 1ml:3000pcs/48.5*40.5*43.5cm 2/3ml:3200pcs/61.5*48.5*38cm 5ml:2400pcs/61.5*48.5*38cm 10ml:2400pcs/61.5*48.5*38cm 20ml:1800pcs/61.5*48.5*38cm 30ml:1200pcs/61.5*48.5*38cm 50/60ml:800pcs/53*47.5.45.5cm
MTP Syringe Product Code: MED 148 Specifications: Sterile, individually packed in peelable blister/HM Pouch pack Size/Strength: Double valve/Single valve Description Manufactured from polypropylene polymer. Specially designed Syringe to apply suction during surgical abortion procedure. MTP Syringe provides High Vacuum Suction during procedure.
Insulin Syringe Product Code: MED 178 Specifications: Sterile, individually packed in Medical Grade Blister & Ribbon Pack Size/Strength: 1ml 40U â?? Insulin, 1ml 100U â?? Insulin, Needle 29G-31G Description Non-Toxic, Non-Pyrogenic and Sterile Single use Latex free needle,Low Dead expose Needle is 20% thinner in diameter (31 G) for lesser penetration force in skin Available in 6mm length and 8mm length
Anti-disable Syringe with needle for Fixed-Dose Immunization With Self-destructive characteristics, the self destructive performance of the syringe will begin to take effects as the injestion starts. When the fixed dose of vaccine is finished, the syringe will be completely sell-locked, effectively preventing the reuse of the syringe and improving the safety of madical injection.
Disposable Insulin Syringe with Needle The integrated design of the needle holder and barrel injection molding which ensures the low residue of the liquid and ensures the accuracy of the injection of the liquid. Excellent sliding performance, convenient for clinical injection operation medical staff.
Insulin Syringe EO Sterilization Refer Standards ISO 9626: 1991 + AMENDMENT 1 2001 Stainless steel needle tubing for the manufacture of medical devices. ISO 8537:2007 Sterile single-use syringes, with or without needle, for insulin. ISO 10993-7:2008 Biological evaluation of medical devices - Part 7 : Ethylene oxide sterilization residuals. ASTM F 1140/F 1140M-13 Standard test methods for internal pressurization failure resistance of unrestrained packages. USP 37-NF 32 Bacterial Endotoxins Limit. ISO 11737-2:2009 Sterilization of medical devices - Microbiological methods - Part 2: Test of sterility performed in the definition, validation and maintenance of a sterilization process. ISO 11135-1:2007 Sterilization of health care products - Ethylene oxide - Part 1: Requirements for development, validation and routine control of a sterilization process for medical devices.